One Human, Multiple Genomes

Faced with questions of personhood and human identity, there is perhaps nothing more unequivocal you could point to than your genome — your personal genetic signature. But what if there isn’t just one genetic signature to which you play host? And what if one or more of those signatures is from another person entirely?
A series of recent studies promises to pile additional layers of complexity onto the already heavily ramified field of genomics. In the NY Times, Carl Zimmer introduces us to evidence that will change how we think about the human genome.
We often think of our cells as specialized entities serving different functions but all housing the same genetic information. It may come as a surprise, then, to know that a cell swabbed from our cheek can have an entirely different genetic code from that of a plucked hair, for example, or from a neuron in the brain. In fact, if you were to sequence a random sample of cells from the same host, you might find a potpourri of genomes, with some even belonging to another person.
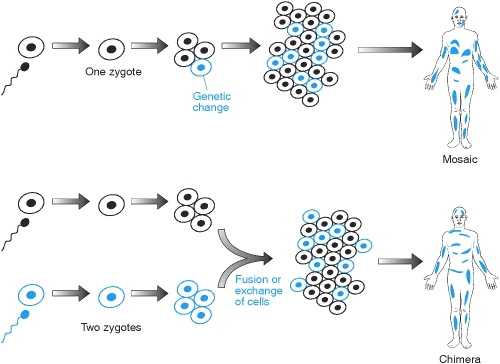
The pair of phenomena associated with these intra-organism variations are formally known as mosaicism and chimerism. We’ve known about the former since the 1930s when American geneticist Curt Stern demonstrated the occurrence through mitosis, a natural cell process utilized by every eukaryotic organism.
When a female egg is fertilized, a single genome — a marital blend of the two parent genomes — emerges. At this stage, there is just one cell with one genome, called a zygote. That cell then divides to form an embryo, but mitosis is an imperfect and infrequently sloppy process. The replicants are often not perfect clones of their parent cells, and genetic dissimilarities can arise. The mutant cells of course also divide, thus birthing the embryo and resulting organism with different sets of genetic instructions. One set may grow into the tissue that forms the lungs, while another may develop into a vital organ.
This scenario is known to occur not only during embryogenesis, but at any stage of a species’ life. Put simply, these mosaic derivatives can pop up at any time as a function of the disorder inherent in various stages of cell division.
Chimerism, by contrast, also occurs in utero but is the result of two or more zygotes swapping cells back and forth in the womb on their way to becoming an embryo. If these cells make mitotic headway in certain locations in the body, twins may share certain cells in common.
Another manner in which a foreign genome can be introduced and spread is from an organ transplant. One study at Innsbruck Medical University found that 74 percent of patients contained the genome of the donor in addition to their own, a full nine years after the transplants were performed.
While initially received with little skepticism, the idea that any cell taken from anywhere in the body will have identical DNA is now just too simplistic a view of biology. Until recently there simply hasn’t been enough original research into this area to conclude how widespread mosaicism and its cognates are.
We have also found that many women harbor cells that contain Y chromosomes. How is this possible? It turns out that genetic material can be passed from fetus to mother. The placenta — nature’s all-in-one life support system — allows for bidirectional exchange of various nutrients and fluids. Embryonic cells can also be left behind after the birth of a child, which can then multiply and assimilate into neighboring tissues.
“It’s pretty likely that any woman who has been pregnant is a chimera,” Dr. Randolph said.”
Indeed, these are not fringe cases which demand unique explanations, but evidenced by several focused studies to date. Zimmer recounts one such study conducted last year.
“In 2012, Canadian scientists performed autopsies on the brains of 59 women. They found neurons with Y chromosomes in 63 percent of them. The neurons likely developed from cells originating in their sons.”
What about blood type? Is it possible to have not one but two different blood types? Indeed it is. Mothers and fetuses can intermingle red blood cells during and after gestation. That means if you’re a mother you may have some Type A cells and some Type O cells, for example. While women who carry fetuses with incongruous blood types normally develop antibodies to suppress them, the mismatched type often lingers in the blood stream long after the baby is born.1
Challenges Ahead
Our newfound view of cellular nonconformity is certain to have rippling import for everyone from geneticists and oncologists to forensic investigators. To be sure, multiple DNA sequences deflate some of the confidence services like 23andMe and other genetic counseling and personal genomics services can provide. Sequencing the genome of a single group of cells with one suite of medical predispositions and physiological baselines doesn’t tell you anything about cells with a different genetic manual.
Medical practitioners will need to work out how the presence of multiple genomes impacts the host. Specifically, we need a better understanding of how isolated blotches of a “foreign” or mutant genome fit into the overall genetic canvas. Is genetic diversity spread across different sites of the body something that promotes proper health, or is this cause for concern?
As you can imagine, such a situation can also create headaches for forensic scientists working to establish identity of victims and criminals.
“Last year, for example, forensic scientists at the Washington State Patrol Crime Laboratory Division described how a saliva sample and a sperm sample from the same suspect in a sexual assault case didn’t match.”
With this and other research into the microbiome, it’s clear that speaking of our genome in the singular is no longer tenable. We are curator to a mosaic of genetic programming, and the implications and links to medical science will take time to fully dissect. If this latest research has shown us anything, it’s that no neat and tidy schema for reality is immune to being ruptured by new evidence.
External link: DNA Double Take
Further reading: You can actually have two sets of DNA
- In rare cases, it is even possible for your blood type to change. As mentioned, an organ transplant can alter the genetic makeup of the recipient, but certain kinds of transplantation can even overwrite the patient’s blood type over time, such as bone marrow transplants. To prevent body-wide rejection, doctors will ideally use a donor that is a match in blood type and other biomarkers, especially human leukocyte antigens (HLA), but once the recipient’s marrow is replaced, it will begin pumping out the donor’s blood cells. It may take several weeks or months for the original blood type to be replaced entirely, but eventually it will. [↩]


Comments